1. Basics of fires
2. Factors and causes of fire
3. How can we reduce the risk of fire
4. Fire tetrahedron
5. Questions and answers
Basics of Fires
The major cause of accident in oil & gas industries is due to fire. There are many flammable & inflammable fluids & solids present in the oil & gas plant which ignites to cause fire.
Let us discuss the factors & causes of fire.
Factors and causes of fire in oil and gas industries-
Fire Triangle
The concept of fire can be well understood and explained using a simple model called the Fire Triangle. As seen in the fire triangle the three factors contributing to cause a fire are: Fuel, Oxidizing agent (Oxygen) & Heat. A fire is caused if all the three factors are present in mixture in the required concentration. Reverse is true for stopping the fire to happen i.e.if any one of the factor is eliminated or the concentration of any one can be kept below the required level then fire can be eliminated.

Fig 1 Fire Triangle
How can we reduce the risk of fire?
Fuel
Typical cases where fuel occurs are – if there is a leak, during filling operations, transfer operations, or excessive dusts.
- Although we often cannot always eliminate these sources we can reduce the possibilities by having good ventilation to keep vapors from building up.
- Often we locate things out-doors, use grating on floors so vapors don’t build up.
Oxidizers
- Oxygen is the most common oxidizer, especially that found in ambient air.
- For oxygen, we often use “inerting” with nitrogen, helium blankets over flammable materials to reduce O2 content below that where you can have combustion.
Ignition Sources
-
- Heat is a common ignition source.
- Ignition sources are plenty
- Although we can eliminate ignition sources, it is almost inevitable that an ignition source will be available if there is a large release of flammable material that cannot be diluted quickly.
Fire Tetrahedron
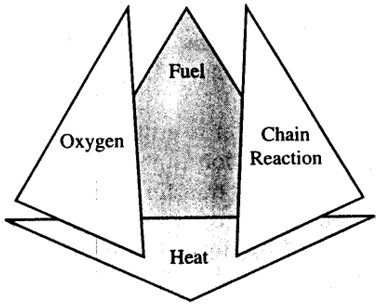
Fire Tetrahedron
In above discussion we have seen that the main factors to cause a fire are: Fuel, Oxidizing agent (Oxygen) & Heat, but the main factor which allows the fire to sustain and continue is the Chemical Chain Reaction. These four factors form a Fire Tetrahedon which allows the fire to continue unless one of the factors is blocked.
Water is one of most important fire retardant agent as it keeps the temperature of the fuel below its ignition point. Similarly foam prevents the required oxygen level for fire to happen.
Explosions
- Detonation – explosion (chemical reaction) with shock wave greater than speed of sound
- Deflagration – explosion (chemical reaction) with shock wave less than speed of sound
- BLEVE – Boiling Liquid Expanding Vapor Explosion – when liquid is at a temperature above its atmospheric boiling point. Vessel ruptures – flammable liquid flashes and results in a fire/explosion
- Confined explosion – an explosion occurring within a vessel or a building. Usually results in injury to the building inhabitants and extensive damage.
- Unconfined explosion – an explosion occurring in the open. Usually results from spill of a flammable gas spill. These explosions are rarer than confined since dilution occurs.
- Dust Explosions – This explosion results from the rapid combustion of fine solid particles. Many solid materials become very flammable when reduced to a fine powder.
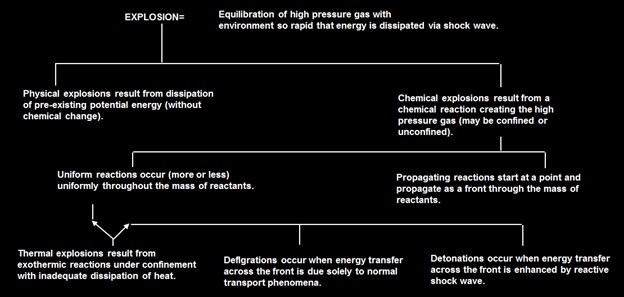
Explosions
You may refer to below related topics-
Types of Fire Protection Systems
Concept and Need for Passive Fire Protection
Few important questions related to Fire & Explosion
Que: What is the flash point of a liquid?
Ans. Flash point is the lowest temperature at which a liquid exposed to the air gives off sufficient vapor to form a flammable mixture, or within the apparatus used, that can be ignited by a suitable flame. More precisely, it is the temperature of a liquid at which the partial pressure of its vapor reaches the lower flammable limit when the liquid is heated in air.
Que: What is the fundamental difference between flammable and combustible stock?
Ans. Flammable stock is capable of being ignited without having to be heated. Combustible material must be heated by some external source in order to be capable of burning.
Que: What is the cut-off point between a “flammable liquid” and a “combustible liquid” as defined by the NFPA standards?
Ans. NFPA defines a flammable liquid as one having a flash point below 100oF (37.8oC). A combustible liquid is one with a flash point of 100oF (37.8oC) or above.
Que: What is the difference between the terms “lower explosive limit (LEL)” and “lower flammable limit (LFL)”?
Ans. None. The terms are synonymous.
Que: A material whose flash point is 212oF (100oC) is being stored at 203oF (95oC). Is this treated as a flammable or combustible material under ExxonMobil practices?
Ans. Flammable (Stored within 15oF (10oC) of its flash point.
Que: There is a correlation of flash point with upper flammable limit (UFL) by means of the vapor pressure curve. (True/False)
Ans. False. The correlation is with the lower flammable limit (LFL).
Que: A pipe whose surface temperature is 662oF (350oC) represents a likely source of ignition for a flammable vapor whose autoignition temperature (A.I.T.) is 608oF (320oC). (True/False).
Ans. False. In order to be a source of ignition in open air, a hot line would have to be at least 220oF (105oC) higher than the AIT. This has been found by experiment. Apparently, natural convection prevents the vapor from remaining in contact with the pipe long enough to cause ignition.
Que: Pressure has a significant effect on the flammable range of most hydrocarbons. (True/False).
Ans. True. Flammable range widens with increasing pressure.
Que: Deflagration is another word for detonation. (True/False)
Ans. False. Deflagration is characterized by sub-sonic flame velocities, whereas detonation shock waves are supersonic.
Que: Typical pressures reached in a confined deflagration are 6 to 8 times the initial pressure. (True/False)
Ans. True.
Que: Stoichiometric mixtures generally require higher ignition energies than other mixtures within the flammable range. (True/False)
Ans. False. They require lower energies.
Que: The only factors that determine the strength of a vapor cloud explosion are the type of molecule and the amount released. (True/False)
Ans. False. Other factors are confinement, weather, and source consideration.
Que: The TNT model is still the best for modeling explosions. (True/False)
Ans. False. Although explosions are still reported as “tons of TNT equivalent”, the Multi-Energy Model is more accurate in most cases.